Crackdown On Combination
 The Government has enacted a sweeping ban on over 300 Fixed Dose Combinations (FDCs). This move has stirred controversy, particularly surrounding the inclusion of Metformin in several banned formulations. Metformin is used to treat high blood sugar levels that are caused by type 2 diabetes…
The Government has enacted a sweeping ban on over 300 Fixed Dose Combinations (FDCs). This move has stirred controversy, particularly surrounding the inclusion of Metformin in several banned formulations. Metformin is used to treat high blood sugar levels that are caused by type 2 diabetes…
By Prof (Dr) Ramesh K Goyal
Recently, the Central Government of India, through Gazette Notifications nos. S.O.4379 (E) to S.O.4706 (E), prohibited 328 Fixed dose combinations (FDCs) for manufacture, sale, or distribution with immediate effect under Section 26A of the Drugs and Cosmetics Act, 1940. This decision was based on the recommendation of the Drugs Technical Advisory Board (DTAB), citing either a lack of therapeutic justification for the active pharmaceutical ingredients (APIs) contained in these combinations or potential risks to human health. Notably, among the banned FDCs, 27 combinations involved Metformin at various dose levels, sparking widespread public attention and controversy. While some companies expressed discontent, several FDCs were subsequently withdrawn from the market.
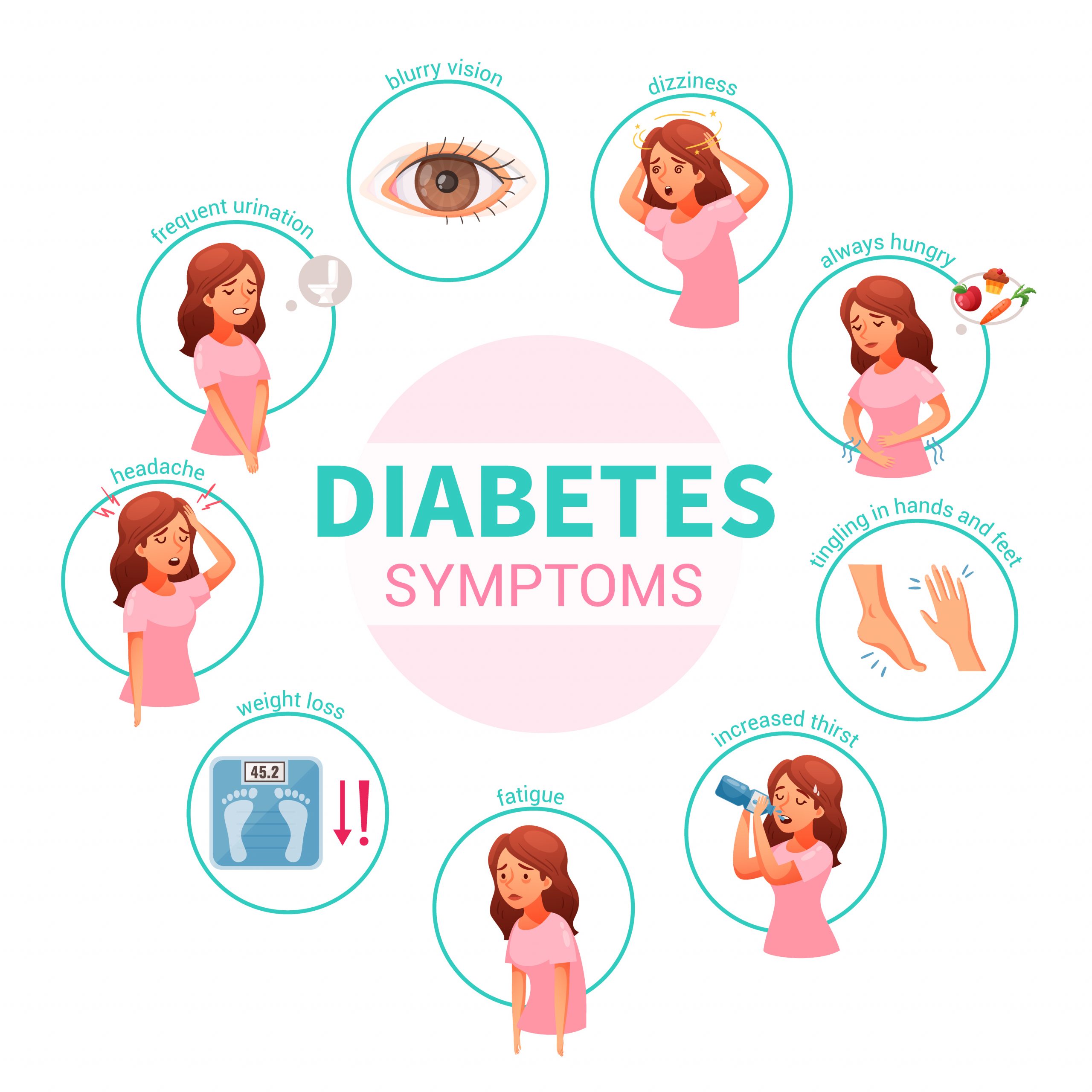
Out of the over 300 banned FDCs, more than 70 were used as painkillers. Some of these painkillers contained antacids or anti-gastric acid secretion medications, which may not be therapeutically necessary. Additionally, many formulations were intended for cold or bronchial asthma, with steroids being a common component. It is well-known that steroids cannot be used for prolonged periods due to the risk of serious adverse effects. Furthermore, some FDCs were so irrational that they included the banned drug itself. While painkillers or cold medications may not be used consistently over extended periods, those intended for chronic conditions like hypertension or diabetes pose a more significant risk, as patients often rely on them for years, necessitating frequent dosage adjustments. This presents a serious challenge.
FDCs are combinations of two or more active pharmaceutical ingredients/drugs in a single dosage form, sometimes referred to as “polypills.” Polypills can be beneficial for patients with multiple coexisting conditions, as they simplify medication regimens. However, FDCs require closer scrutiny due to the potential for drug-drug interactions and stability concerns. While FDCs may be cheaper to manufacture and distribute, they have some disadvantages compared to single drug formulations.


The rational use of FDCs in the management of type 2 diabetes mellitus requires careful evaluation of both therapeutic efficacy and safety considerations. The recent ban on several FDCs highlights the importance of regulatory oversight in ensuring the quality and safety of medications used in diabetes management.
The proliferation of Fixed Dose Combinations (FDCs) in the Indian pharmaceutical market has raised concerns both domestically and internationally regarding their safety, efficacy, and regulation. While FDCs can offer benefits such as improved adherence and enhanced therapeutic effects, their rationality must be carefully evaluated to ensure patient safety.
In response to these concerns, regulatory bodies in many countries, including India, have implemented measures to oversee the development and marketing of FDCs. However, challenges persist, particularly in a market where thousands of FDCs are available, ranging from safe and effective formulations to those deemed unsafe or dangerous.

The World Health Organization (WHO) has provided guidelines for the registration of fixed-dose medicinal products, aimed at guiding countries lacking established regulatory frameworks for such products. These guidelines serve as a reference for ensuring the rationality and safety of FDCs in various healthcare settings.
FDCs are often utilised in the treatment of infectious diseases like HIV, malaria, and tuberculosis, where multiple antimicrobial agents are required for optimal efficacy. Additionally, they play a role in managing chronic conditions characterised by multiple co-existing disorders.
The rationality of FDCs hinges on several key aspects, including the mechanisms of action of the drugs in the combination, pharmacokinetic compatibility, and the absence of supra-additive toxicity. FDCs deemed safe and effective based on these criteria are widely used in clinical practice, while those deemed unsafe or dangerous are subject to regulatory scrutiny and potential restrictions.
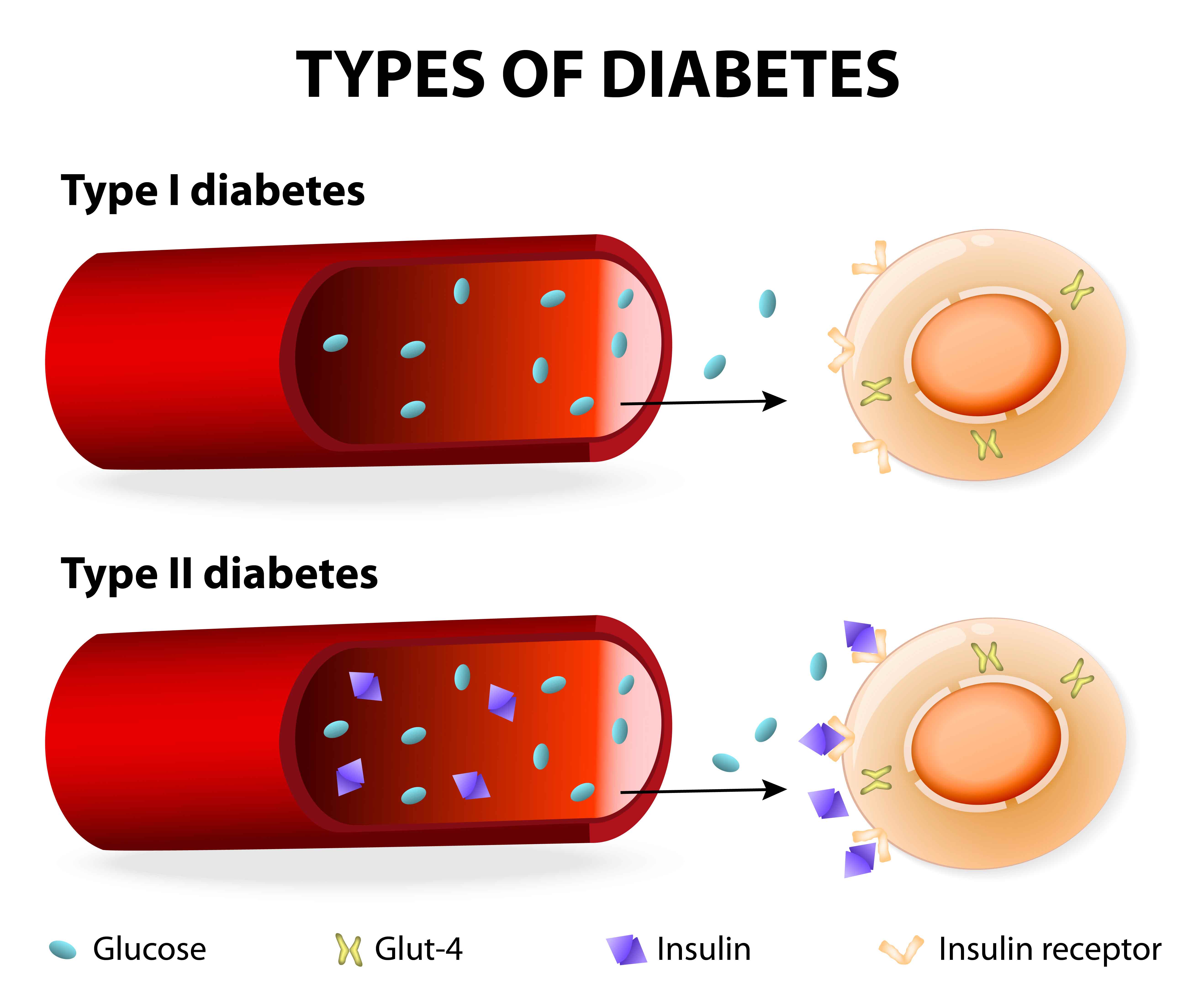
Rationale for Fixed Dose Combinations
An FDC must demonstrate that it is rational to combine two or more APIs into a single product. Therefore, the rationality should depend on quality, medical, and bioavailability considerations.
Quality: The same quality standards that apply to single-component products will apply to FDCs. It will be necessary to demonstrate that the quality of the combination is similar to that of the individual ingredients.
Medical: There should be a medical rationale for combining the APIs, such as increased efficacy compared to the individual components given at the same dose, similar pharmacokinetics, and similar duration of action. Additionally, the incidence of adverse reactions in response to treatment with the combination should be lower than that in response to any of the component actives given alone. Apart from these, FDCs might show dose reduction, cost reduction, improved adherence, and simplified therapy.
Bioavailability considerations: Interpretation of the results of bioavailability (BA) and bioequivalence (BE) tests involves both quality and medical considerations. For example, it is not acceptable if the bioavailability of the FDC is reduced or variable compared with that of single-entity products due to poor formulation. However, an interaction between two actives that leads to increased bioavailability may be one of the advantages considered when balancing advantages and disadvantages.
Irrational Fixed Dose Combinations: The most pressing concern with irrational FDCs is that they expose patients to unnecessary risks of adverse drug reactions. Irrational FDCs also impose an unnecessary financial burden on consumers. Many irrational FDC medicines have been approved by state and central regulatory authorities in India, and their use is promoted extensively by pharmaceutical firms. There is growing national and international concern about the drug regulatory system in India. Parliamentary reports have highlighted the presence of a high number of unapproved medicines and irrational combinations of both approved and unapproved drugs in the Indian marketplace. FDCs are a peculiar feature of the Indian pharmaceutical landscape.
FDCs and Type 2 Diabetes Mellitus: In India, more than 60 million people have been diagnosed with type 2 diabetes mellitus, which has been described as ‘the diabetes capital of the world.’ In a recent review published elsewhere, authors have mentioned that apart from constant monitoring and rapid adjustment of treatment regimens required to maintain adequate glycaemia control, metformin-FDCs are not recommended by national or international treatment guidelines. Nevertheless, the Central Drugs Standard Control Organization (CDSCO), India’s drug regulator, has given approval for 52 FDC formulations for type 2 diabetes, which in turn has given rise to more than 500 marketed brands of metformin FDCs. In contrast, only two metformin FDCs are approved in the USA, one in Australia, and none in either the UK or Canada. Twenty-seven metformin-FDCs were included in the Indian government’s ban of 344 unapproved FDCs in March 2016 as having ‘no therapeutic justification’ following consideration of nearly 6000 unapproved FDCs by an expert committee. The ban was lifted in December 2016 by the Delhi High Court and was upheld by the Supreme Court in December 2017. The Supreme Court required the Drugs Technical Advisory Board to consider the banned drugs (except those approved before 1988) and to submit a report within 6 months. Recently, based on the recommendation of DTAB, the Central Govt. of India via Gazette Notifications nos. S.O.4379(E) to S.O.4706(E) dated 7th September 2018, has prohibited all the 27 metformin-FDCs for manufacture, sale, or distribution with immediate effect under Section 26A of the Drugs and Cosmetics Act, 1940.
While FDCs offer potential benefits in improving treatment outcomes and patient adherence, their rationality must be carefully evaluated to mitigate risks to patient safety. Regulatory oversight and adherence to established guidelines are essential in ensuring the quality, efficacy, and safety of FDCs in the Indian pharmaceutical market and beyond.
(The author is Former Vice Chancellor, Delhi Pharmaceutical Sciences and Research University, New Delhi)

